Original title: Impact of Chemical Properties of Human Respiratory Droplets
and Aerosol Particles on Airborne Viruses’ Viability and Indoor Transmission
Source link: https://msystems.asm.org/content/5/2/e00245-20#ref-list-1
Published: July 2022
1. Vejerano, E.P.; Marr, L.C. Physico-chemical characteristics of evaporating respiratory fluid droplets. J. R. Soc. Interface 2018,
15, 20170939. [CrossRef]
2. Niazi, S.; Groth, R.; Spann, K.; Johnson, G.R. The role of respiratory droplet physicochemistry in limiting and promoting the
airborne transmission of human coronaviruses: A critical review. Environ. Poll. 2021, 276, 115767. [CrossRef] [PubMed]
3. Huynh, E.; Olinger, A.; Woolley, D.; Kohli, R.K.; Choczynski, J.M.; Davies, J.F.; Lin, K.; Marr, L.C.; Davis, R.D. Evidence for a
semi-solid phase state of aerosols and droplets relevant to the airborne and surface survival of pathogens. Proc. Nat. Acad. Sci.
USA 2022, 119, e2109750119. [CrossRef] [PubMed]
4. Liu, L.; Wei, J.; Li, Y.; Ooi, A. Evaporation and dispersion of respiratory droplets from coughing. Indoor Air 2017, 27, 179–190.
[CrossRef] [PubMed]
5. Sosso, G.C.; Chen, J.; Cox, S.J.; Fitzner, M.; Pedevilla, P.; Zen, A.; Michaelides, A. Crystal nucleation in liquids: Open questions
and future challenges in molecular dynamics simulations. Chem. Rev. 2016, 116, 7078–7116. [CrossRef]
6. Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.;
Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N. Engl. J. Med.
2020, 382, 1564–1567. [CrossRef]
7. Prather, K.A.;Wang, C.C.; Schooley, R.T. Reducing transmission of SARS-CoV-2. Science 2020, 368, 1422–1424. [CrossRef]
8. Asadi, S.; Bouvier, N.; Wexler, A.S.; Ristenpart, W.D. The coronavirus pandemic and aerosols: Does COVID-19 transmit via
expiratory particles? Aerosol. Sci. Technol. 2020, 54, 635–638. [CrossRef]
9. Morawska, L.; Cao, J. Airborne transmission of SARS-CoV-2: The world should face the reality. Environ. Int. 2020, 139, 105730.
[CrossRef]
10. Bukhari, Q.; Jameel, Y. Will Coronavirus Pandemic Diminish by Summer? SSRN, 2020; Available online: https://papers.ssrn.com/
sol3/papers.cfm?abstract_id=3556998 (accessed on 1 January 2022).
11. Ahlawat, A.;Wiedensohler, A.; Mishra, S.K. An Overview on the role of relative humidity in airborne transmission of SARS-CoV-2
in indoor environments. Aerosol. Air Qual. Res. 2020, 20, 1856–1861. [CrossRef]
12. Moriyama, M.; Hugentobler, W.J.; Iwasaki, A. Seasonality of respiratory viral infections. Ann. Rev. Virol. 2020, 7, 83–101.
[CrossRef] [PubMed]
13. Lin, K.; Schulte, C.R.; Marr, L.C. Survival of MS2 and F6 viruses in droplets as a function of relative humidity, pH, and salt,
protein, and surfactant concentrations. PLoS ONE 2020, 15, e0243505. [CrossRef]
14. Fedorenko, A.; Grinberg, M.; Orevi, T.; Kashtan, N. Survival of the enveloped bacteriophage Phi6 (a surrogate for SARS-CoV-2) in
evaporated saliva microdroplets deposited on glass surfaces. Sci. Rep. 2020, 10, 22419. [CrossRef] [PubMed]
15. Beggs, C.B.; Avital, E.J. A psychrometric model to assess the biological decay of the SARS-CoV-2 virus in aerosols. PeerJ 2021,
9, e11024. [CrossRef] [PubMed]
16. Niazi, S.; Groth, R.; Cravigan, L.; He, C.; Tang, J.W.; Spann, K.; Johnson, G.R. Susceptibility of an airborne common cold virus to
relative humidity. Environ. Sci. Technol. 2021, 55, 499–508. [CrossRef]
17. Niazi, S.; Short, K.R.; Groth, R.; Cravigan, L.; Spann, K.; Ristovski, Z.; Johnson, G.R. Humidity dependent survival of an airborne
Influenza A virus: Practical implications for controlling airborne viruses. Environ. Sci. Technol. Lett. 2021, 8, 412–418. [CrossRef]
18. Benbough, J.E. Some factors affecting the survival of airborne viruses. J. Gen. Virol. 1971, 10, 209–220. [CrossRef]
19. Johnson, G.R.; Morawska, L. The mechanism of breath aerosol formation. J. Aerosol. Med. Pulm. Drug Deliv. 2009, 22, 229–237.
[CrossRef]
20. Bozic, A.; Kanduc, M. Relative humidity in droplet and airborne transmission of disease. J. Biol. Phys. 2021, 47, 1–29. [CrossRef]
21. Effros, R.M.; Hoagland, K.W.; Bosbous, M.; Castillo, D.; Foss, B.; Dunning, M.; Gare, M.; Lin, W.E.; Sun, F. Dilution of respiratory
solutes in exhaled condensates. Am. J. Respir. Crit. Care Med. 2002, 165, 663–669. [CrossRef]
22. Yang, W.; Elankumaran, S.; Marr, L.C. Concentrations and size distributions of airborne influenza A viruses measured indoors at
a health centre, a day-care centre and on aeroplanes. J. R. Soc. Interface 2011, 8, 1176–1184. [CrossRef] [PubMed]
23. Nicas, M.; Nazaroff,W.W.; Hubbard, A. Towards understanding the risk of secondary infection: Emission of respirable pathogens.
J. Occup. Environ. Hyg. 2005, 2, 143–154. [CrossRef] [PubMed]
24. Reynolds, H.Y.; Chrétien, J. Respiratory tract fluids: Analysis of content and contemporary use in understanding lung diseases.
Disease-a-month 1984, 30, 1–103. [CrossRef]
25. Ali, M.; Lillehoj, E.P.; Park, Y.; Kyo, Y.; Kim, K.C. Analysis of the proteome of human airway epithelial secretions. Proteome Sci.
2011, 9, 4. [CrossRef]
26. Schenkels, L.C.; Veerman, E.C.; Nieuw Amerongen, A.V. Biochemical composition of human saliva in relation to other mucosal
fluids. Crit. Rev. Oral. Biol. Med. 1995, 6, 161–175. [CrossRef] [PubMed]
27. Kormuth, K.A.; Lin, K.; Prussin, A.J.; Vejerano, E.P.; Tiwari, A.J.; Cox, S.S.; Myerburg, M.M.; Lakdawala, S.S.; Marr, L.C. Influenza
Virus Infectivity Is Retained in Aerosols and Droplets Independent of Relative Humidity. J. Infect. Dis. 2018, 218, 739–747.
[CrossRef]
28. Kormuth, K.A.; Lin, K.; Qian, Z.; Myerburg, M.M.; Marr, L.C.; Lakdawala, S.S. Environmental Persistence of Influenza Viruses Is
Dependent upon Virus Type and Host Origin. mSphere 2019, 4, e00552-19. [CrossRef]
29. Lin, K.; Marr, L.C. Humidity-dependent decay of viruses, but not bacteria, in aerosols and droplets follows disinfection kinetics.
Environ. Sci. Technol. 2020, 54, 1024–1032. [CrossRef]
30. Pica, N.; Bouvier, N.M. Environmental factors affecting the transmission of respiratory viruses. Curr. Opin. Virol. 2012, 2, 90–95.
[CrossRef]
31. Yang,W.; Marr, L.C. Mechanisms by which ambient humidity may affect viruses in aerosols. Appl. Environ. Microbiol. 2012, 78,
6781–6788. [CrossRef]
32. Song, M.; Ham, S.; Andrews, R.J.; You, Y.; Bertram, A.K. Liquid–liquid phase separation in organic particles containing one and
two organic species: Importance of the average O: C. Atmos. Chem. Phys. 2018, 18, 12075–12084. [CrossRef]
33. Setti, L.; Passarini, F.; De Gennaro, G.; Barbieri, P.; Perrone, M.G.; Borelii, M.; Palmisani, J.; Di Gilio, A.; Piscitelli, P.; Miani, A.
Airborne transmission route of COVID-19: Why 2 meters/6 feet of inter-personal distance could not be enough. Int. J. Environ.
Res. Public Health 2020, 17, 2932. [CrossRef] [PubMed]
34. Freedman, M.A. Phase separation in organic aerosol. Chem. Soc. Rev. 2017, 46, 7694–7705. [CrossRef]
35. Benton, D.J.; Gamblin, S.J.; Rosenthal, P.B.; Skehel, J.J. Structural transitions in influenza haemagglutinin at membrane fusion pH.
Nature 2020, 583, 150–153. [CrossRef]
36. Dasika, G.K.; Letchworth, G.J. Homologous and heterologous interference requires bovine herpesvirus-1 glycoprotein D at the
cell surface during virus entry. Microbiology 2000, 81, 1041–1049. [CrossRef] [PubMed]
37. Gilbert, J.M.; Mason, D.I.A.N.E.; White, J.M. Fusion of Rous sarcoma virus with host cells does not require exposure to low pH.
J. Virol. 1990, 64, 5106–5113. [CrossRef]
38. Lakadamyali, M.; Rust, M.J.; Babcock, H.P.; Zhuang, X. Visualizing infection of individual influenza viruses. Proc. Nat. Acad. Sci.
USA 2003, 100, 9280–9285. [CrossRef]
39. Stiasny, K.; Heinz, F.X. Flavivirus membrane fusion. J. Gen. Virol. 2006, 87, 2755–2766. [CrossRef]
40. White, J.; Kartenbeck, J.; Helenius, A. Fusion of Semliki forest virus with the plasma membrane can be induced by low pH. J. Cell
Biol. 1980, 87, 264–272. [CrossRef]
41. Wang, H.; Yang, P.; Liu, K.; Guo, F.; Zhang, Y.; Zhang, G.; Jiang, C. SARS coronavirus entry into host cells through a novel
clathrin-and caveolae-independent endocytic pathway. Cell Res. 2008, 18, 290–301. [CrossRef]
42. Nenes, A. Exploring the Science of Humidity and Its Impact on Viruses, Their Hosts, and Human Well-Being (10 May 2021).
Available online: https://smw.ch/expert-webinars/indoor-air-and-covid-transmission (accessed on 9 January 2022).
43. Imai, M.; Watanabe, T.; Hatta, M.; Das, S.C.; Ozawa, M.; Shinya, K.; Zhong, G.; Hanson, A.; Katsura, H.; Watanabe, S.; et al.
Experimental adaptation of an influenza H5 HA confers respiratory droplet transmission to a reassortant H5 HA/H1N1 virus in
ferrets. Nature 2012, 486, 420–428. [CrossRef] [PubMed]
44. Hemmes, J.; Winkler, K.; Kool, S. Virus survival as a seasonal factor in influenza and poliomyelitis. Antonie Van Leeuwenhoek 1962,
28, 221–233. [CrossRef] [PubMed]
45. Hemmes, J.H.;Winkler, K.; Kool, S.M. Virus survival as a seasonal factor in influenza and poliomyelitis. Nature 1960, 188, 430–431.
[CrossRef] [PubMed]
46. Harper, G.J. Airborne micro-organisms: Survival tests with four viruses. J. Hyg. 1961, 59, 479–486. [CrossRef]
47. Shechmeister, I.L. Studies on the experimental epidemiology of respiratory infections III. Certain aspects of behavior of type A
influenza virus as an airborne cloud. J. Infect. Dis. 1950, 87, 128–132. [CrossRef]
48. Schaffer, F.L.; Soergel, M.E.; Straube, D.C. Survival of airborne influenza virus: Effects of propagating host, relative humidity, and
composition of spray fluids. Arch. Virol. 1976, 51, 263–273. [CrossRef]
49. Chan, K.H.; Peiris, J.S.M.; Lam, S.Y.; Poon, L.L.M.; Yuen, K.Y.; Seto, W.H. The effects of temperature and relative humidity on the
viability of the SARS coronavirus. Adv. Virol. 2011, 2011, 134690. [CrossRef]
50. Darnell, M.E.R.; Subbarao, K.; Feinstone, S.M.; Taylor, D.R. I nactivation of the coronavirus that induces severe acute repiratory
syndrome, SARS-CoV. J. Virol. Methods 2004, 121, 85–97. [CrossRef]
51. Michen, B.; Graule, T. Isoelectric points of viruses. J. Appl. Microb. 2010, 109, 388–397. [CrossRef]
52. Duran-Meza, A.L.; Villagrana-Escareño, M.V.; Ruiz-Garcı’a, J.; Knobler, C.M.; Gelbart,W.M. Controlling the surface charge of
simple viruses. PLoS ONE 2021, 16, e0255820.
53. Heffron, J.; Mayer, B.K. Virus isoelectric point estimation: Theories and methods. Appl. Environ. Microb. 2021, 87, e02319-20.
[CrossRef] [PubMed]
54. Joonaki, E.; Hassanpouryouzband, A.; Heldt, C.L.; Areo, O. Surface Chemistry Can Unlock Drivers of Surface Stability of
SARS-CoV-2 in a Variety of Environmental Conditions. Chem 2020, 6, 2135–2146. [CrossRef] [PubMed]
55. Ault, A.P.; Axson, J.L. Atmospheric aerosol chemistry: Spectroscopic and microscopic advances. Anal. Chem. 2017, 89, 430–452.
[CrossRef] [PubMed]
56. Pye, H.O.T.; Nenes, A.; Alexander, B.; Ault, A.P.; Barth, M.C.; Clegg, S.L.; Fahey, K.M.; Hennigan, C.J.; Herrmann, H.; Kanakidou,
M.; et al. The acidity of atmospheric particles and clouds. Atmos. Chem. Phys. 2020, 20, 4809–4888. [CrossRef] [PubMed]
57. Almstrand, A.C.; Ljungstrom, E.; Lausmaa, J.; Bake, B.; Sjovall, P.; Olin, A.C. Airway monitoring by collection and mass
spectrometric analysis of exhaled particles. Anal. Chem. 2009, 81, 662–668. [CrossRef]
58. Bredberg, A.; Gobom, J.; Almstrand, A.C.; Larsson, P.; Blennow, K.; Olin, A.C.; Mirgorodskaya, E. Exhaled endogenous particles
contain lung proteins. Clin. Chem. 2012, 58, 431–440. [CrossRef]
59. McDevitt, J.J.; Koutrakis, P.; Ferguson, S.T.;Wolfson, J.M.; Fabian, M.P.; Martins, M.; Pantelic, J.; Milton, D.K. Development and
Performance Evaluation of an Exhaled-Breath Bioaerosol Collector for Influenza Virus. Aerosol. Sci. Technol. 2013, 47, 444–451.
[CrossRef]
60. Identifying the Coronavirus in One Breath. Available online: https://ch.ambafrance.org/Das-Coronavirus-in-einem-Atemzugerkennen
(accessed on 9 January 2022).
61. Ahlawat, A.; Mishra, S.K.; Birks, J.W.; Costabile, F.; Wiedensohler, A. Preventing Airborne Transmission

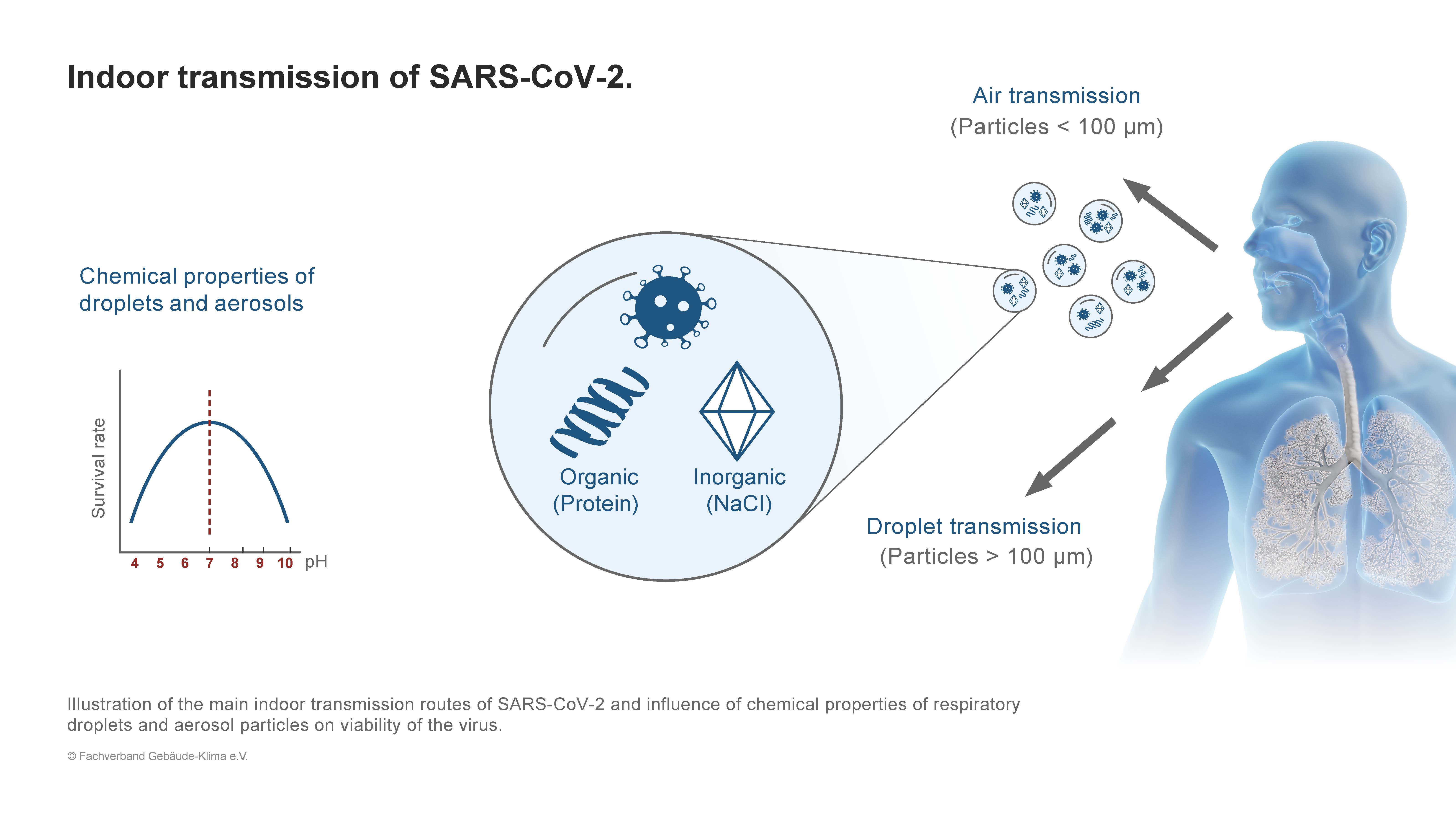 Figure 1
Figure 1 Figure 2
Figure 2 2.2.3. Virus Isoelectric Points and Their Dependence on pH
2.2.3. Virus Isoelectric Points and Their Dependence on pH