What is isothermal humidification?
The term ‘isothermal humidification’ has become established to describe a method of humidification using water vapour in which the air temperature remains essentially constant during the humidification process.
Depending on the system, steam humidification can be controlled with a high degree of precision and, due to the use of a medium at over 100 °C, offers excellent hygiene standards. A steam humidifier can be powered by electricity or gas.
How a steam humidifier works
Unlike evaporative humidifiers, the water vapour is generated before being mixed with air. The vapour can then be fed directly into the room air via a ventilation unit or mixed into the air within an air-conditioning system during the conditioning process. The steam is either drawn from a central steam generation system or generated locally at the point of air humidification for air conditioning.
When air is humidified using pressurised steam from a central source, the term ‘pressurised steam humidifiers’ is used. This type of steam humidifier is used in conjunction with HVAC systems (heating, ventilation and air conditioning systems). The humidifier consists of a dirt trap, a rotary valve, a pressure gauge and a condensate drain.
If the steam is to be generated directly at the point of use, electric steam humidifiers are often a sensible choice.
An electrically operated steam air humidifier works either on the electrode or resistance principle. In electrode humidifiers, grid-shaped metal electrodes are immersed in the water tank and utilise the conductivity of the water. The current flows directly through the water, causing it to evaporate. In this system, the steam output is regulated by the water level in the steam cylinder. In a resistance-type steam humidifier, the water is heated using the immersion heater principle.
The different types of steam
For isothermal air humidification, water must first be heated to 100°C. At this temperature, it has a heat capacity of 419 kJ/kg and a pressure of 1.0133 bar. At this point, a change in the state of matter occurs, whereby boiling water at 100°C is converted into steam at 100°C. In the process, 2,258 kJ of energy must be supplied per kg of water.
The energy required for this conversion into steam is referred to as the specific heat of vaporisation. This is latent, meaning it cannot be measured with a thermometer.
Saturated steam
As long as steam is in direct contact with the water, it is ‘saturated’, meaning it cannot absorb any more liquid and is therefore also referred to as ‘saturated steam’.
Unsaturated steam
Unsaturated steam is produced when the volume of the steam increases, but the temperature is kept constant despite the supply of heat. An important characteristic here is the condensate. When steam is cooled, the heat of vaporisation invested in it is removed, its state of matter changes and condensate is formed.
Formation of limescale during evaporation
Why does limescale form?
Calcium is the fifth most abundant element in the Earth’s crust. It is very often not found in its pure form, but rather combined with other substances, such as limestone, or dissolved in water. Limestone consists mainly of calcium carbonate (CaCO3).
By determining the water hardness, the intensity of limescale formation can also be predicted Water hardness is a conceptual framework in applied chemistry that has developed from the practical needs of using natural water with its dissolved constituents. Specifically, water hardness refers to the equivalent concentration of the ions of the alkaline earth metals dissolved in water, but in specific contexts also to their anionic partners. The ‘hardness-forming elements’ essentially include calcium and magnesium, as well as traces of strontium and barium. The dissolved hardness-forming elements can form insoluble compounds, primarily limescale and so-called lime soaps. This tendency to form insoluble compounds is the reason for the attention that has led to the development of the conceptual and theoretical framework surrounding water hardness.
Soft water is more suitable for all applications where the water is heated, for washing, for watering houseplants, etc. Soft water is available in regions with granite, gneiss, basalt and schist. Rainwater is also soft water.
Hard water leads to limescale build-up in household appliances, increases the consumption of washing-up liquid and detergent, and affects the taste and appearance of delicate foods and drinks (e.g. tea). Hard water comes from regions where sandstone and limestone predominate.
Solution to the limescale problem
To reduce limescale build-up in steam-air humidification, Condair’s patented limescale management system can be used, whereby limescale deposits are blasted off the heating elements and drained via an external limescale collection tank.
Furthermore, fully demineralised water can be used, from which minerals and calcium have already been removed through water treatment. The amount of limescale that then remains is negligible.
It should be noted, however, that operation with softened (i.e. mineral-free) water is not possible with every type of humidifier. A resistance humidifier can also be operated with mineral-free water. An electrode humidifier, on the other hand, requires conductive, mineral-rich water to operate efficiently. Operating this type of humidifier with softened water is not energy-efficient.
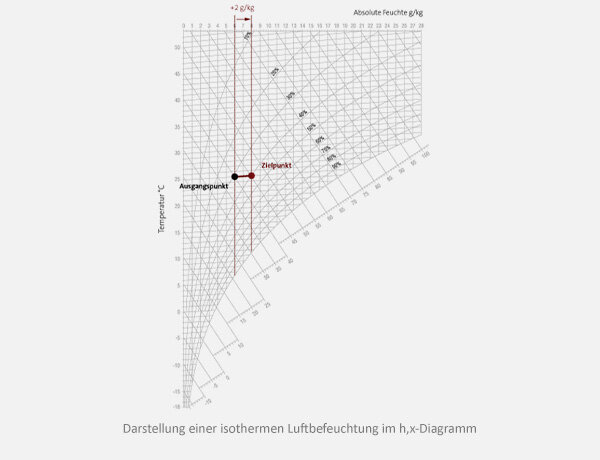
Calculation example: Isothermal air humidification
Given:
Calculation of ∆h/∆x:
∆h=kJ/kg
∆x =g/kg dry air
Get in touch with us
We look forward to receiving your message and your needs for Condair solutions.
Here you can obtain further information or contact your Condair representatives in your region directly.
Need more information or have a question?
Contact us via our contact form
Contact a Condair consultant directly
Find the right Condair contact persons in your region